
Wisconsin Academy of sciences arts & Letter
People & Ideas: Spring 2012
A recent proposal to extract mineral resources from the Penokee Range, a low mountain ridge in Northern Wisconsin containing a huge deposit of iron-rich rocks, has stirred up great controversy around jobs, environmental protection, politics, and the rights of sovereign native nations.
The controversy began when Gogebic Taconite LLC, (GTAC) a Florida-based mining company, indicated interest in mining a four-mile stretch of Penokee Range bedrock running through Iron and Ashland counties from just south of Hurley to the little town of Mellen, Wisconsin. GTAC announced plans to create a 1.5 billion-dollar mine over the next thirty years that would produce approximately 14 million tons of iron ore per year. Too, GTAC claimed they would bring to the economically depressed Northwest region of the state hundreds of jobs.
With a mandate from Governor Scott Walker to create jobs and GTAC eager to set up operations in Wisconsin, the State Assembly passed a mining reform bill (AB 426) in January 2012 to speed the permitting process for iron mines. The legislation made iron-mining regulations separate from those that govern the mining of other metals and loosened many of the existing environmental regulations on the industry. A similar bill, proposed to the Wisconsin State Senate in early March 2012, was narrowly defeated (17–16) in a rancorous session.
Later that month GTAC suspended their pursuit of an open-pit mine in the Penokee Range.
As with any precious metal, the price of iron ore fluctuates according to market demand. Even though GTAC dropped the project, they or another company—for better or worse—might again pursue the rights to mine the Penokee Range. While the immediate future of mining legislation in Wisconsin is uncertain, the iron-rich rocks are not going away—and neither are the complex issues surrounding metals mining.
A FASCINATING GEOLOGIC HISTORY
Something that you don’t hear much about in discussions for or against the proposed mining of the Penokee Range, is the unique geologic history and composition of the rocks found therein—the examination of which I believe is central to a balanced and informed perspective on the issue of mining in Wisconsin.
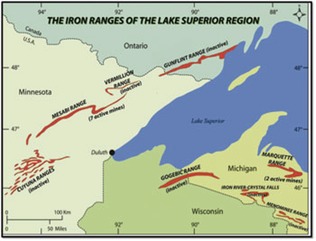
Our examination starts with the band of iron-rich rock, called the Ironwood Iron Formation, that forms the backbone of the Penokee Range. The Ironwood extends 75 miles from Lake Gogebic in the Upper Peninsula of Michigan (where the Penokee is called the Gogebic Range) to just south of Grand View, Wisconsin (see Figure 1). The hardness of this formation and its resistance to erosion is the main reason the Penokee Range stands tall above the surrounding less-resistant rocks.
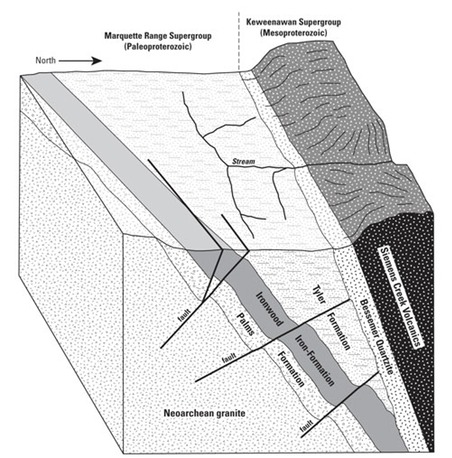
About 480-feet thick and sandwiched between a quartz-rich layer called the Palms Formation on the southern side and the predominantly slate Tyler Formation to the north (see Figure 2), the Ironwood has a fascinating geologic history and a distinctly different mineral composition than that of the other six bedrock formations found in the range.
Iron formations like this are unusual in that they formed abundantly during only one period of Earth history that ended very abruptly 1.85 billion years ago. These sedimentary formations are somewhat of an enigma because there is no place on Earth where geologists can observe similar deposits being created today. Therefore, everything about their origin has to be inferred from geologic clues preserved in the rocks. Specifically, their layering and sedimentary grains suggest some of the layers were deposited in shallow water influenced by waves, whereas other layers were deposited just offshore below the waves at a time when the sea level was higher.
The deposition of these rocks took place about 1.9 billion years ago. Continents were scattered across the globe in different places than today, and they occupied much less of the Earth’s surface. Earth’s atmosphere was very different, too: rich in carbon dioxide and methane, but very poor in oxygen. Seawater was also poor in oxygen, which meant that iron could be dissolved at much higher concentrations than is possible in today’s comparatively oxygen-rich oceans. Around this time, photosynthesis by algae was just starting to take place on a large scale, causing a slight increase in the oxygen concentration in the atmosphere and oceans. This slight increase was just enough to allow the dissolved iron to react with oxygen and precipitate out of the water, creating the iron oxide minerals hematite and magnetite— the same minerals we find today in the distinctive Ironwood Iron Formation.
After a few hundred million years during which iron-rich sediments were deposited in abundance on the ocean floor, the conditions that made this possible suddenly changed. Why did conditions change? Geologists working in the Lake Superior region have recently discovered that iron formation deposition likely ceased due to an enormous meteorite impact at what is now Sudbury, Ontario. It is thought that the force of the impact created massive tsunamis that caused the Earth’s oceans to roil and mix, changing seawater chemistry in such a way that iron formations could no longer be deposited. The top of the Ironwood Formation, and corresponding iron formations in Michigan and Minnesota, are all marked by a distinctive layer of broken rock that records the shock wave and tsunami created by the meteorite impact.
A UNIQUE MINERAL COMPOSITION
The layers of sedimentary rock found in the Ironwood Iron Formation, originally deposited in a horizontal fashion, were tipped steeply to the north at about 65 degrees by tectonic forces acting on the region about 1.1 billion years ago. Today, just the edges of the formations are at or near the Earth’s surface, offering us but a glimpse of the ancient conditions and tremendous forces that brought them to where they are. But this glimpse can tell us a lot. Composed of thin layers of the minerals quartz (SiO2) and magnetite (Fe3O4)—an iron oxide—in varying proportions, the Ironwood Formation is a relatively rich deposit of iron ore.
Iron ores are iron oxide-rich rocks and minerals from which metallic iron can be economically extracted. The iron itself in these rocks and minerals—which vary in color from dark grey, bright yellow, deep purple, to rusty red—is usually found in the form of magnetite, hematite, goethite, limonite, or siderite.
Magnetite is the mineral of interest to mining companies in this case because it contains a lot of ferrous (Fe+2) iron—levels so high that a magnet is attracted to this mineral. Indeed, there are areas of the Penokee Range where the rock is so rich in iron that a compass will point toward the rock rather than point north. This magnetic property also makes it relatively simple to extract the magnetite from crushed ore rock using large magnets, a process central to mining formations like the kind found in the Penokee Range.
In some areas of the Ironwood Formation there are abundant layers of quartz, constituting a rock that is commonly referred to as banded iron formation, or BIF. These areas don’t have the same potential economic value as the layers of taconite, the term for a low-grade ore rock rich enough in iron minerals like magnetite to make it worthwhile to mine for profit (see Figure 3).
The economic and resource importance of the Ironwood Formation has been well known for a long time. About forty mines worked the Ironwood Formation between 1877 and 1967, when more than 325 million tons of ore were extracted from mines located between Upson, Wisconsin, and Wakef ield, Michigan. Most of this ore came from underground workings where soft, highgrade ore with very high quantities of magnetite (greater than ~60% iron) was extracted through narrow shafts and tunnels. These high-grade ores are known as natural ore or direct-shipping ore, meaning they can be fed directly into iron-making blast furnaces with no processing.
Eventually these underground mines were abandoned when the natural ore became scarce. Today, the remaining natural ore of the Penokee Range is in pockets too small to be economically mined. But the grade of taconite ore found in the range is quite good, with 25% to 30% of the weight of the rock made up of iron.
New mining technology—including the development of giant mining machines that can extract hard taconite ore from open pit mines—allows for the extraction of up to 200 tons of rock out of the mine in a single load. Too, refined methods of processing bring down the overall cost of extracting the iron from the taconite pellets. The large amount of remaining taconite together with the recent high price and demand for iron has again made the Ironwood attractive to some mining companies.
Of course, the grade of the ore is only one of many factors that determine whether a deposit is viable. Total size of the deposit is another important factor, and the volume of the deposit in the proposed GTAC site near Mellen is extremely large. It has been estimated that the Ironwood Formation contains 3.7 billion tons of economic ore, making it one of the largest iron reserves in North America. That quantity of ore could be turned into about one billion tons of steel. For comparison, the three-foot-diameter main cables of the Golden Gate Bridge are each 1.5 miles long and contain 12,250 tons of steel. Thus, the economic ore of the Penokee Range is the equivalent of 120,000 miles of three-foot-diameter steel cable. This is enough three-foot cable for about 40,000 Golden Gate Bridges, or enough cable to wrap around the Earth almost five times.
Between 1995 and 2008 the United States used on average 56 million tons of iron ore per year. At that rate of use, the Ironwood Formation contains 66 years of domestic supply. Of course, there are large deposits remaining in other iron ranges in Minnesota and Michigan, so it is likely that ore wouldn’t be extracted from only one iron range at a time. The GTAC plan called for mining around 14 million tons of ore per year over a period of 30 years, an amount equal to about 25% of the iron ore used per year in the U.S. The demand for iron will probably be high for decades, at least, so the apparent departure of GTAC from Wisconsin does not signal the end of interest in the Penokee Range.
IMPACTS OF MINING
While every geologic setting is different, studies of existing mines elsewhere show that iron mining comes with physical, chemical, and biological impacts to the land. Some of these impacts are immediately apparent, while others take a while to manifest. Obvious and immediate physical changes include large-scale modification of Earth’s surface resulting from the removal of huge volumes of rock and the storage of large amounts of waste rock. A large open-pit mine in the Penokee Range, similar to the one proposed by GTAC, would be about four miles long, 600 to 900 feet deep, and about 1600 feet wide, making it the largest single topographic feature in Wisconsin.
Putting aside these topographic considerations for the moment, one of the primary reasons for the controversy surrounding the proposed mine in northern Wisconsin is the reality of the environmental impacts to the headwaters of the fairly pristine and sensitive ecosystem of the Bad River Watershed. The site of the proposed mine is in the headwaters of Tyler Forks River, which flows into the Bad River at Copper Falls State Park (see Figure 4).
If a mine were developed here, headwater streams of the Tyler Forks would be greatly affected. Stream flow would likely be impacted because of changes in the land surface and redirection of groundwater, which would have to be pumped to lower the water table in order for the mine to operate. Pumping would certainly draw down the water table in the area, so wells close to the mine would have less water than today, or even dry up completely. The hydrogeology is not well understood though, so the extent of impacts on groundwater—like many of the environmental impacts that come with mining—are uncertain.
Some creeks on the south side of the Penokee Range would likely disappear because their valleys would be filled by tailings. Additionally, the amount of suspended sediment clouding the water (referred to as turbidity) could increase because of changes in stream flow and increased supply of loose sediment in the rivers.
Although the iron concentration in the Ironwood Formation is high in comparison to many other iron deposits, there is still a lot of material that is not economic and would therefore end up as waste. This poses one of the major environmental issues surrounding any proposed mine because a lot of problems can arise from the massive piles of waste rock left behind. About 30% of the volume of the Ironwood Formation would end up as steel and the rest would end up as waste, also referred to as tailings. Tailings consist of the quartz that remains after the taconite is crushed down to extremely small particles in order get at the magnetite. These small quartz particles are susceptible to erosion and transportation by wind and flowing water.
Another source of tailings would be the uneconomic zones called interburden or waste-in-ore within the Ironwood Formation, which would have to be removed and put aside in order for the miners to gain access to the taconite ore sandwiched between two other formations. Tailings would also be generated from the bedrock sitting above the Ironwood Formation. Waste rock that sits above an ore deposit is called overburden.
In this case, the overburden—tilted at the same 65-degree angle as the ironrich rock—is a wedge of slate and fine quartzite called the Tyler Formation. As the open pit is being enlarged during mining, a progressively increasing volume of the Tyler Formation would have to be removed for every volume of ore rock removed. The total volume of waste rock from these sources would be in the range of 600 million cubic yards—a mountain of waste rock about 600 feet tall if spread over one square mile—by the time the four-mile-long crevasse was fully mined.
Mining can also have environmental impacts far down-gradient of the mine due to changes in the chemical composition of water flowing from mine pits and tailings piles. Of particular concern is the potential for water to interact with soluble minerals from the mine, dissolving metals that can impact water quality. The two dominant minerals in the Ironwood Formation, magnetite and quartz, are nearly insoluble and as such do not influence the chemistry of water they contact. But there are other minerals present in the rock, including small amounts of pyrite (FeS2), or iron sulfide, which can affect water quality. There is also pyrite present in the Tyler Formation, some of which would end up in the tailings as well. When pulverized and put in contact with oxygen and water at the Earth’s surface, pyrite and other sulfide minerals can undergo chemical reactions that create sulfuric acid. This acid can leach harmful metals and compounds that end up in groundwater and surface water.
It is also possible that sulfate ions released during the weathering of pyrite would affect the growth of wild rice and other elements of the sensitive ecosystem found downstream from the mine. Wild rice is considered to be an indicator of good water quality because it is especially sensitive to changes in the concentration of sulfate ions. The wild rice ecosystem in the lower reaches of the Bad River is among the largest in the Great Lakes region and is ecologically and culturally invaluable, so degradation of the ecosystem would be devastating. Wild rice is central to Ojibwe culture and lifestyle and the Bad River Ojibwe Tribe has worked hard to protect the wetlands ecosystem. As a sovereign nation, the tribe has legal authority to regulate water quality of the Bad River. Their water quality standards will influence what can and will happen in the headwaters of the river.
The amount of pyrite in the Tyler Formation and the presence of other minerals that might act to enhance or reduce the formation of acid are very important factors when we consider the potential impacts of mining. The legislation passed in January 2012 by the Wisconsin State Assembly would have decreased the rigor required in scientific studies regarding potential impacts, making assessment of potential damages difficult. At the same time it would weaken many environmental regulations that protect the Bad River and its tributaries from significant water quality changes. The whole idea of differentiating iron mining from other hard-rock mining is based on the assumption that hazardous minerals do not exist in or adjacent to iron ore deposits. But pyrite is known to exist in both the Ironwood and Tyler Formations, so the whole premise behind the legislation is flawed.
At this point in time we still need scientific studies to assess the basic physical and chemical setting of the Penokee Range and how it might be impacted by mining. The potential impacts on the flow and quality of groundwater and surface water are among the many important questions that need to be addressed. We can obtain reasonably accurate answers to these questions through the right scientific studies of the rocks, the groundwater, the surface water, and the biological communities that live in those environments. These issues need to be well understood so that the potential environmental impacts can be assessed and minimized if a mine is to be developed.
COMPLEX ISSUES SURROUNDING MINING
The recent controversy over mining in Wisconsin just underscores the need for informed and engaged discussion when it comes to issues that affect the entire state. Certainly mining provides jobs and materials necessary for society, but it can also be extremely environmentally damaging, with impacts that last indefinitely (at least on the human time scale). As our global population grows—along with opportunities for people across the world to lead the resource-intensive lifestyles of the wealthier countries— the need for mining will continue far into the future. Yet with our increased understanding of the environmental impacts of mining and improved technological controls to limit the impact of mine wastes, mining today can be less damaging than at any point in our history.
Additionally, a better understanding of ecosystem sensitivities allows scientists to identify areas that are too sensitive to risk any type of mining. The Bad River Watershed, with its vast wetland systems, may be such an ecosystem. What the impacts might be and how they can be prevented will only be determined through rigorous scientific studies. Therefore, mining regulations—whether in Wisconsin or elsewhere—must ensure that adequate environmental impact studies are done in advance. These studies must be based on hard science and open to public scrutiny if we are to ensure that mine wastes will be handled responsibly from start to finish.
Although metals are non-renewable resources, they are infinitely recyclable; which means that future generations can benefit from the resources extracted from mines opened today. However, the benefit comes only if mining is done in an environmentally responsible manner and waste metal is recycled into new products. It is critical that laws protect the environment and yet do not preclude all mining. The mining industry operates on a global scale, as does its search for prospective sites. To not permit mines in one area can lead to the development of even more damaging mines in areas with little or no regulation. Hopefully, someday human population numbers will stabilize and we will be so good at recycling that everyone will have enough resources to have a decent lifestyle and no metals will end up wasted. Perhaps then we will stop mining metals.
Until that time, which may be decades or centuries away, mining must be done—and done in a responsible manner. Wisconsin legislators are in a position to take a lead role in thoughtful legislation that balances the need for jobs, resources, and a clean environment. The goal in Wisconsin and elsewhere should be to show leadership in setting responsible standards for mining and environmental protection, not to speed the development of mines for short-term economic gain at the cost of long-term environmental damage.
Tom Fitz
Tom Fitz earned his BS, MS, and PhD degrees in geology with specialties in bedrock geology and earth history. Tom has been teaching at Northland College in Ashland, Wisconsin, for twelve years, and he enjoys taking students into the field to explore rocks in the Lake Superior Region and in the Rocky Mountains of Wyoming. His passion is reading...
